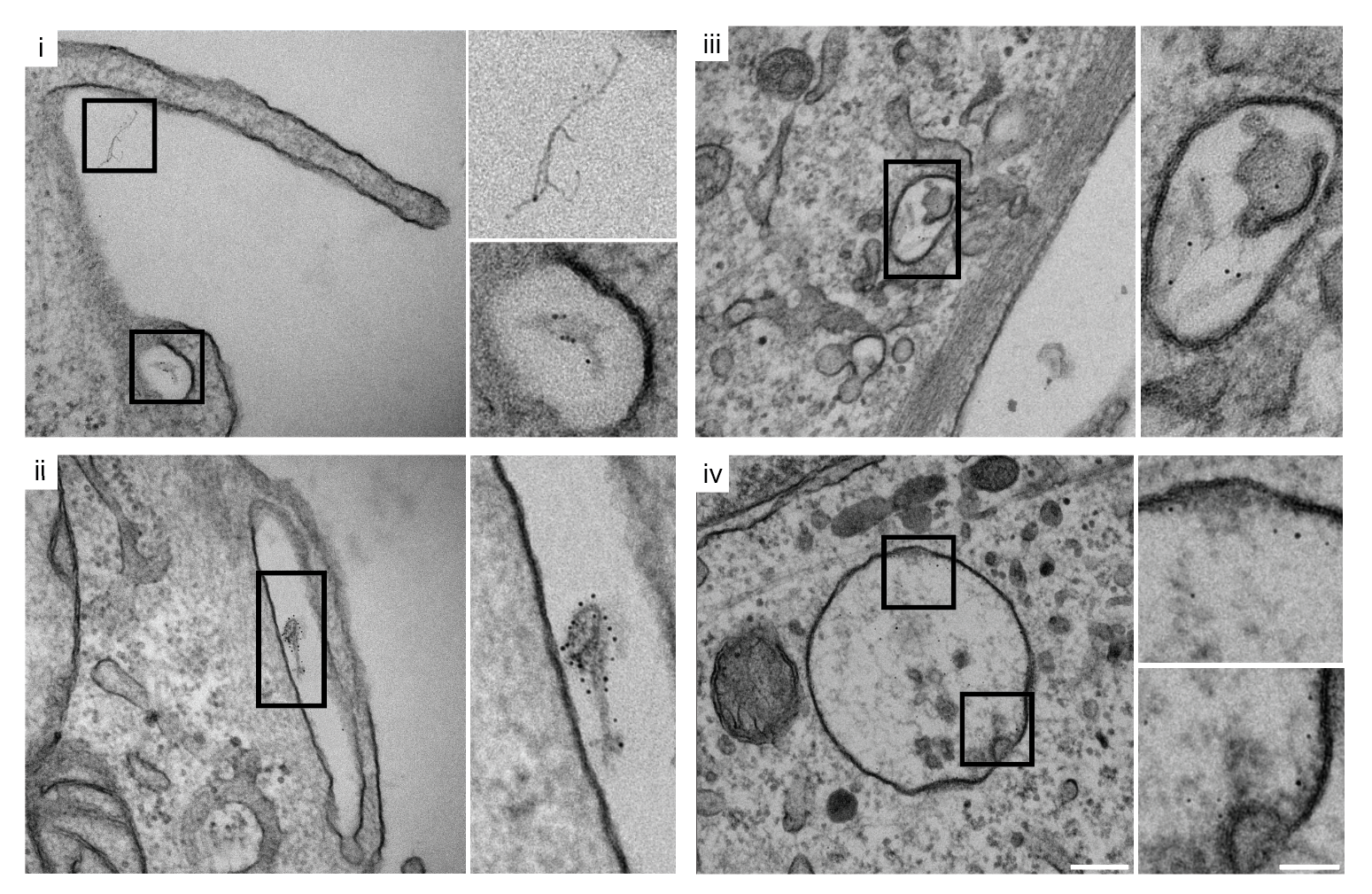
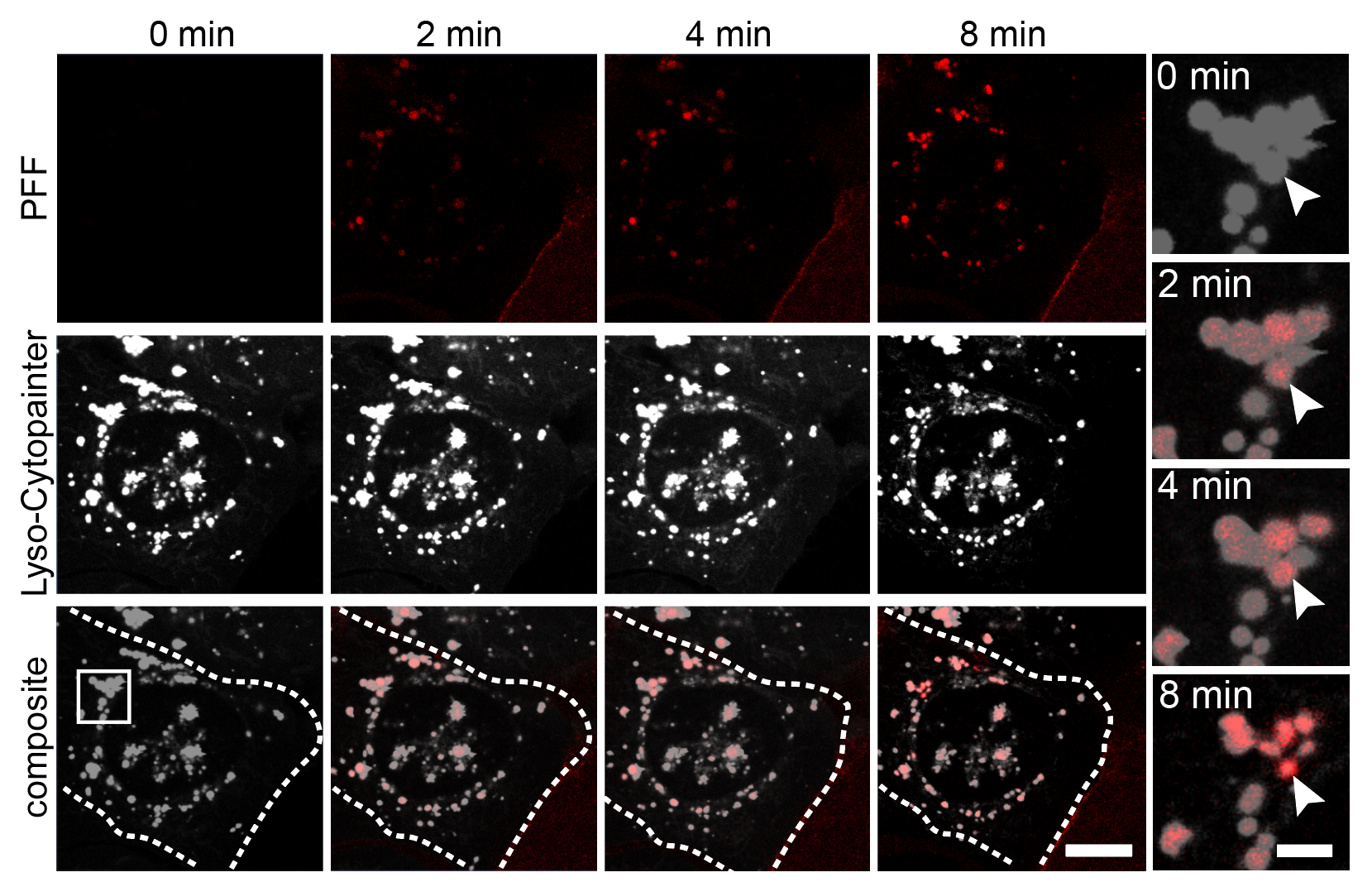
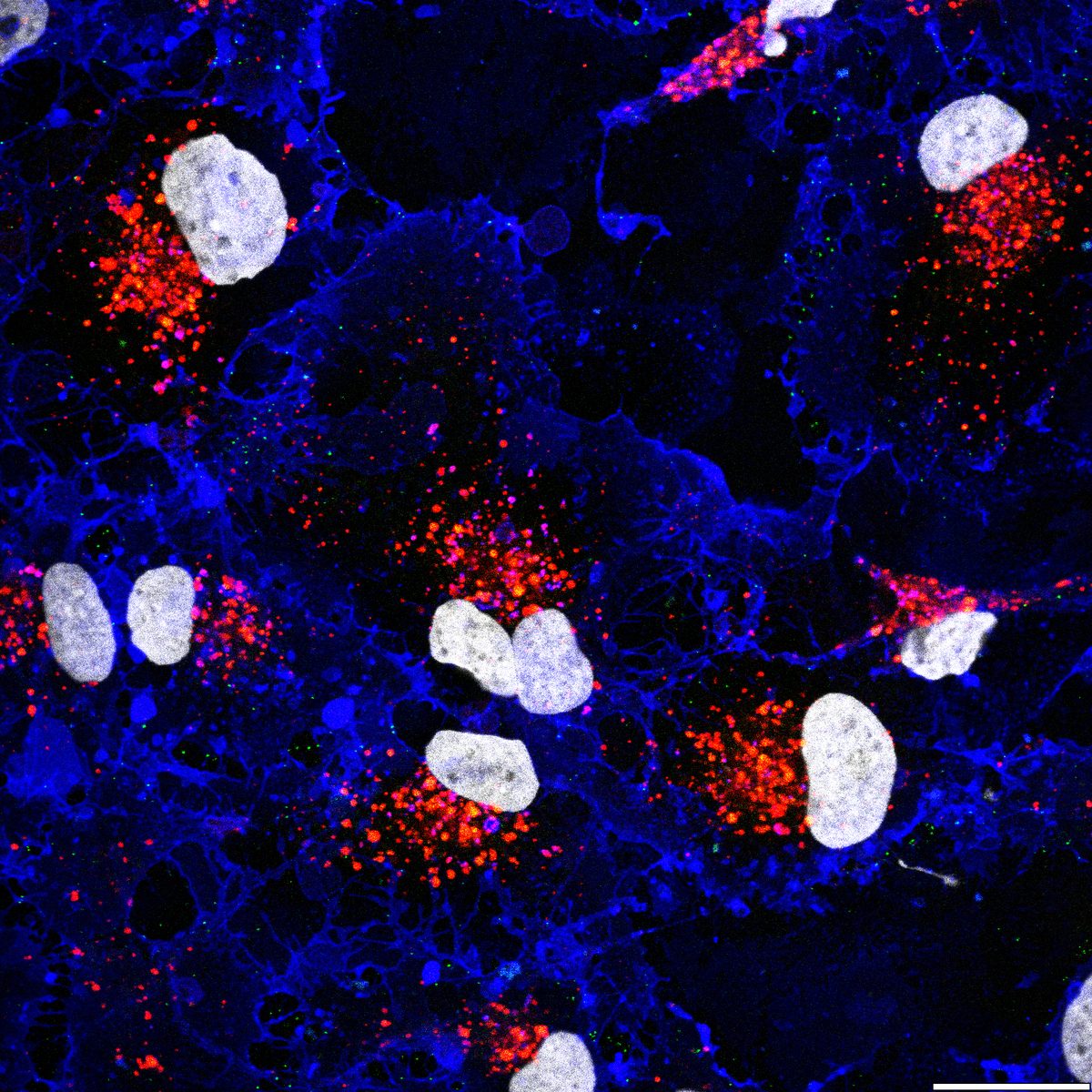
Clathrin-Mediated Endocytosis — Viral RNA Entry via the Endosomal System
Using purified SARS-CoV-2 spike glycoprotein, lentiviral pseudotyping, and live-cell confocal microscopy, I demonstrated that SARS-CoV-2 relies on clathrin-mediated endocytosis (CME) for cell entry — establishing that viral RNA delivery to the cytosol occurs from within the endosomal lumen. This work is directly relevant to understanding how nucleic acid cargoes exploit endocytic machinery to access intracellular compartments, and how disrupting CME can modulate cargo delivery efficiency.
Experimental Approach
Cells expressing ACE2 (HEK-293T-ACE2, Vero, Calu-3) were incubated with purified His6-tagged spike protein or spike-pseudotyped lentivirus. Uptake was tracked by confocal microscopy at defined time points (2, 5, 15, 30 min). CME dependence was assessed via clathrin heavy chain (CHC) siRNA knockdown (~95% KD efficiency), pharmacological inhibition (Pitstop 2, Dynasore), and colocalization with clathrin/AP-2 markers.
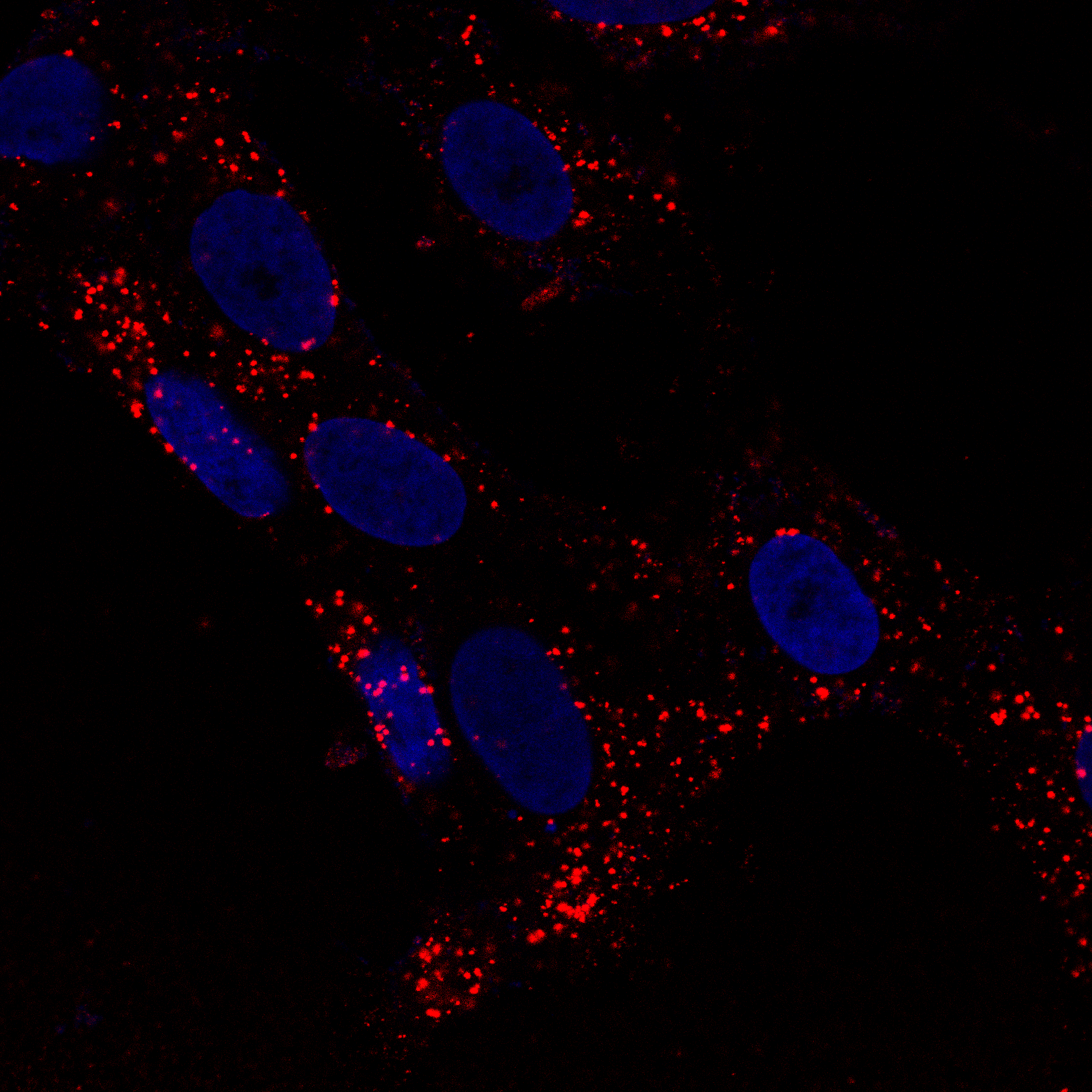
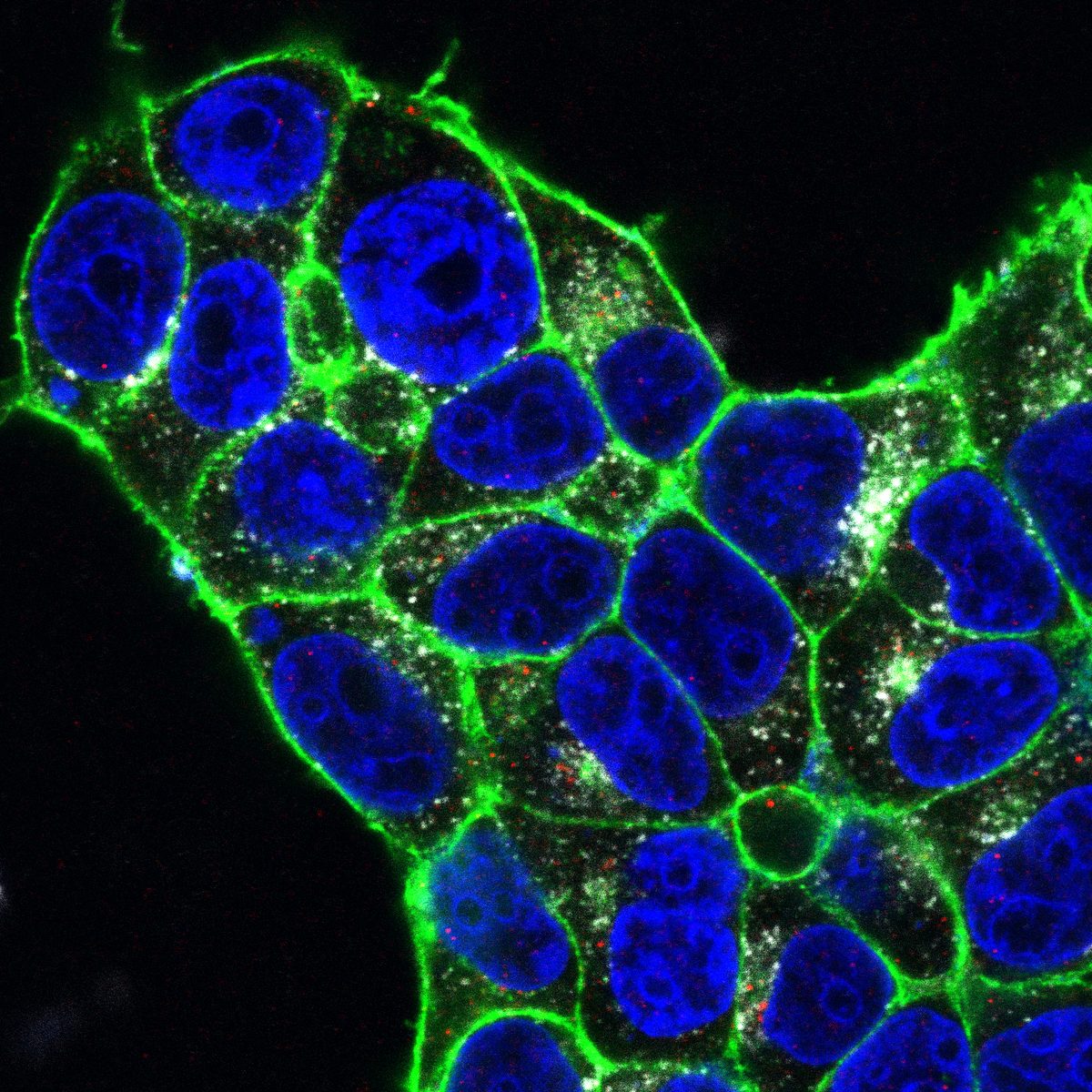
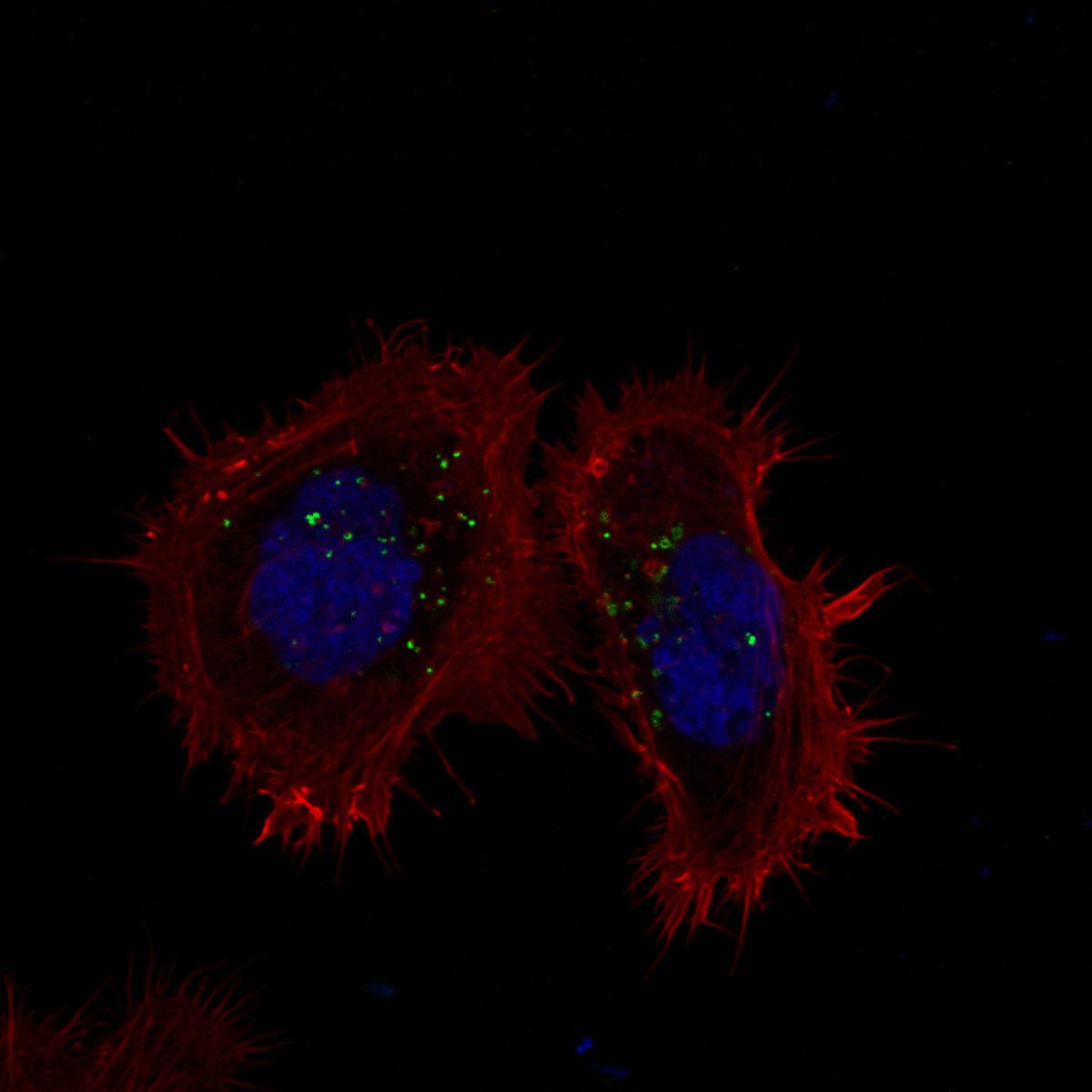
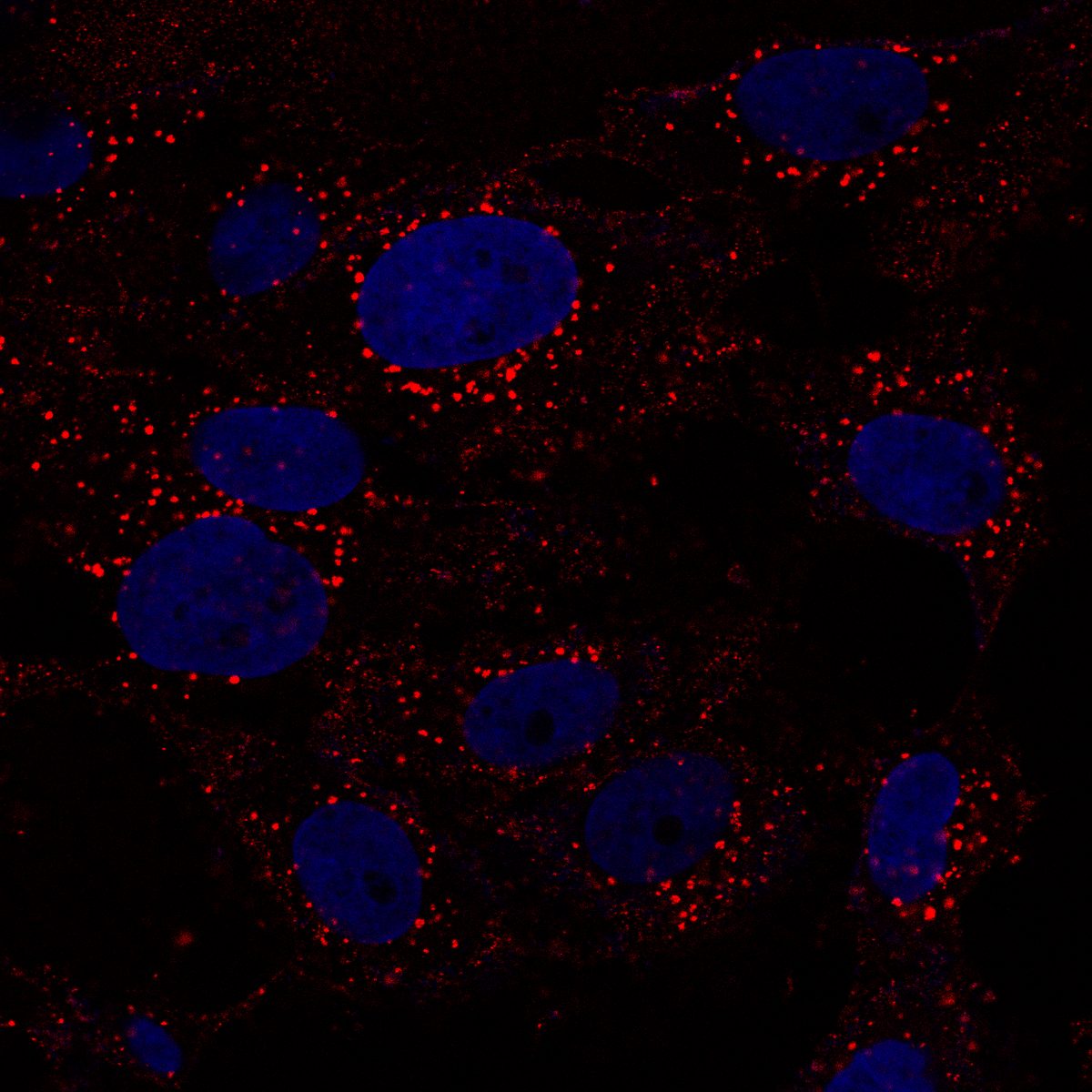
Quantitative CME Internalization Data
Data represent mean ± SEM from 3 independent experiments, n ≥ 200 cells per condition. Spike internalization quantified by intracellular fluorescence intensity normalized to surface-bound signal.
| Condition | Cell Line | Spike Uptake (% Control) | CHC Coloc. (Pearson's R) | Pseudoviral Infectivity (%) |
|---|---|---|---|---|
| Vehicle control | HEK-293T-ACE2 | 100 ± 6.2 | 0.78 ± 0.04 | 100 ± 5.8 |
| CHC siRNA (95% KD) | HEK-293T-ACE2 | 28.4 ± 4.1 | 0.12 ± 0.03 | 18.6 ± 3.2 |
| Vehicle control | Vero | 100 ± 7.4 | 0.74 ± 0.05 | 100 ± 6.1 |
| CHC siRNA (95% KD) | Vero | 31.2 ± 5.3 | 0.14 ± 0.04 | 22.1 ± 4.7 |
| Pitstop 2 (30 μM) | Vero | 42.6 ± 6.8 | 0.21 ± 0.06 | 35.4 ± 5.9 |
| Dynasore (80 μM) | Vero | 38.1 ± 5.5 | 0.18 ± 0.05 | 29.7 ± 4.3 |
| Vehicle control | Calu-3 | 100 ± 8.1 | 0.71 ± 0.06 | 100 ± 7.2 |
| Vehicle control | Caco-2 | 100 ± 5.9 | 0.76 ± 0.04 | 100 ± 4.8 |
Bayati, Kumar, Francis & McPherson. SARS-CoV-2 infects cells following viral entry via clathrin-mediated endocytosis. J. Biol. Chem. 2021; 296:100306.