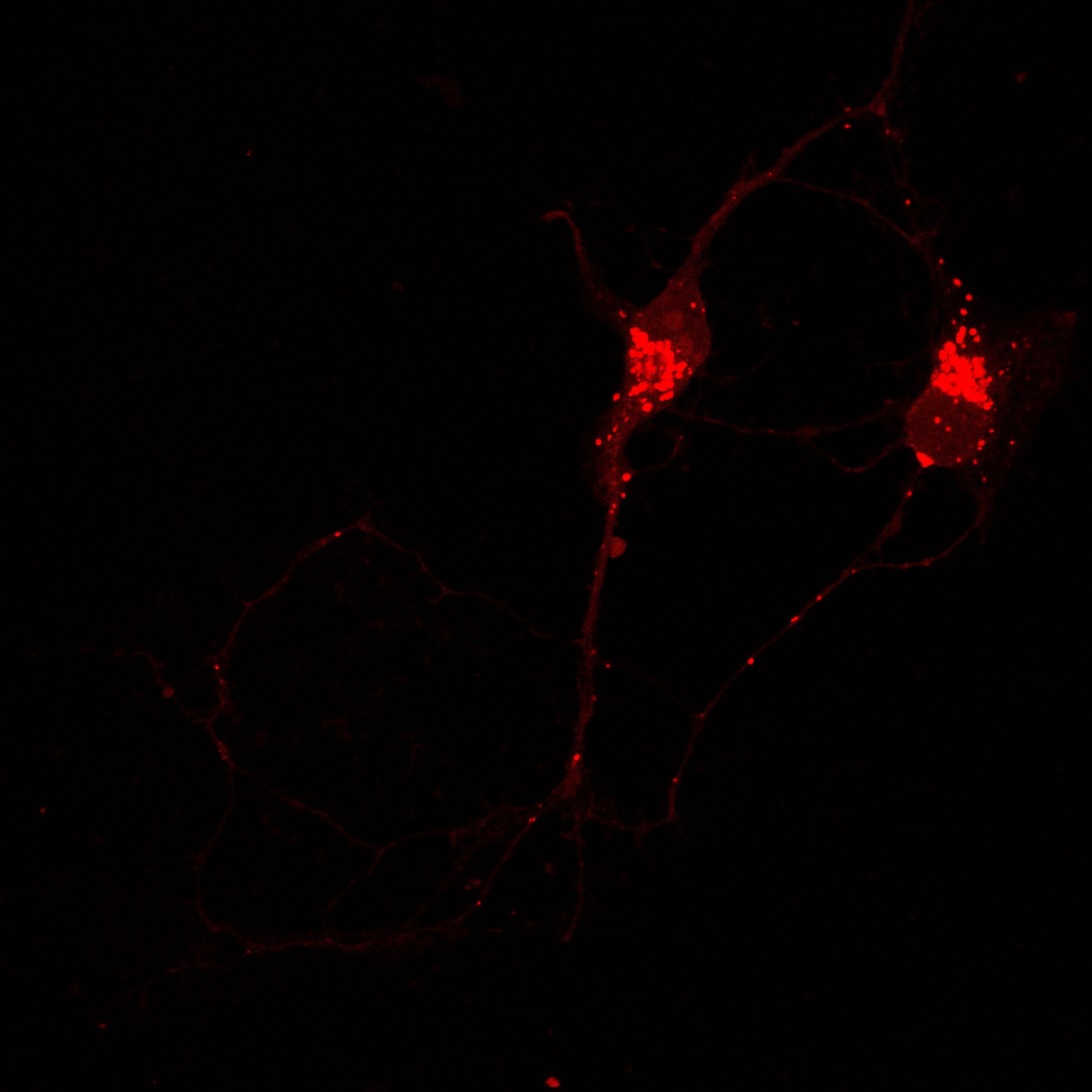
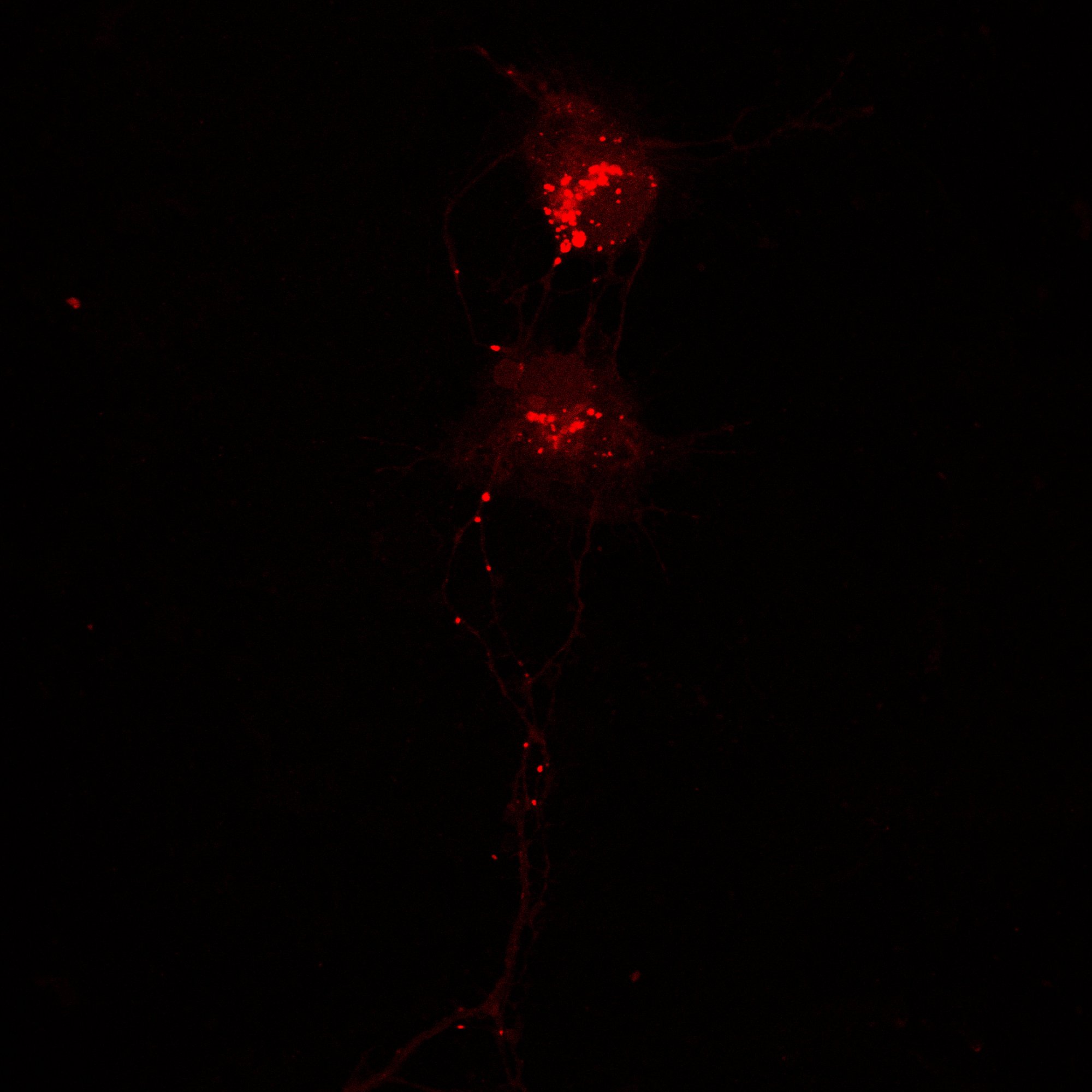
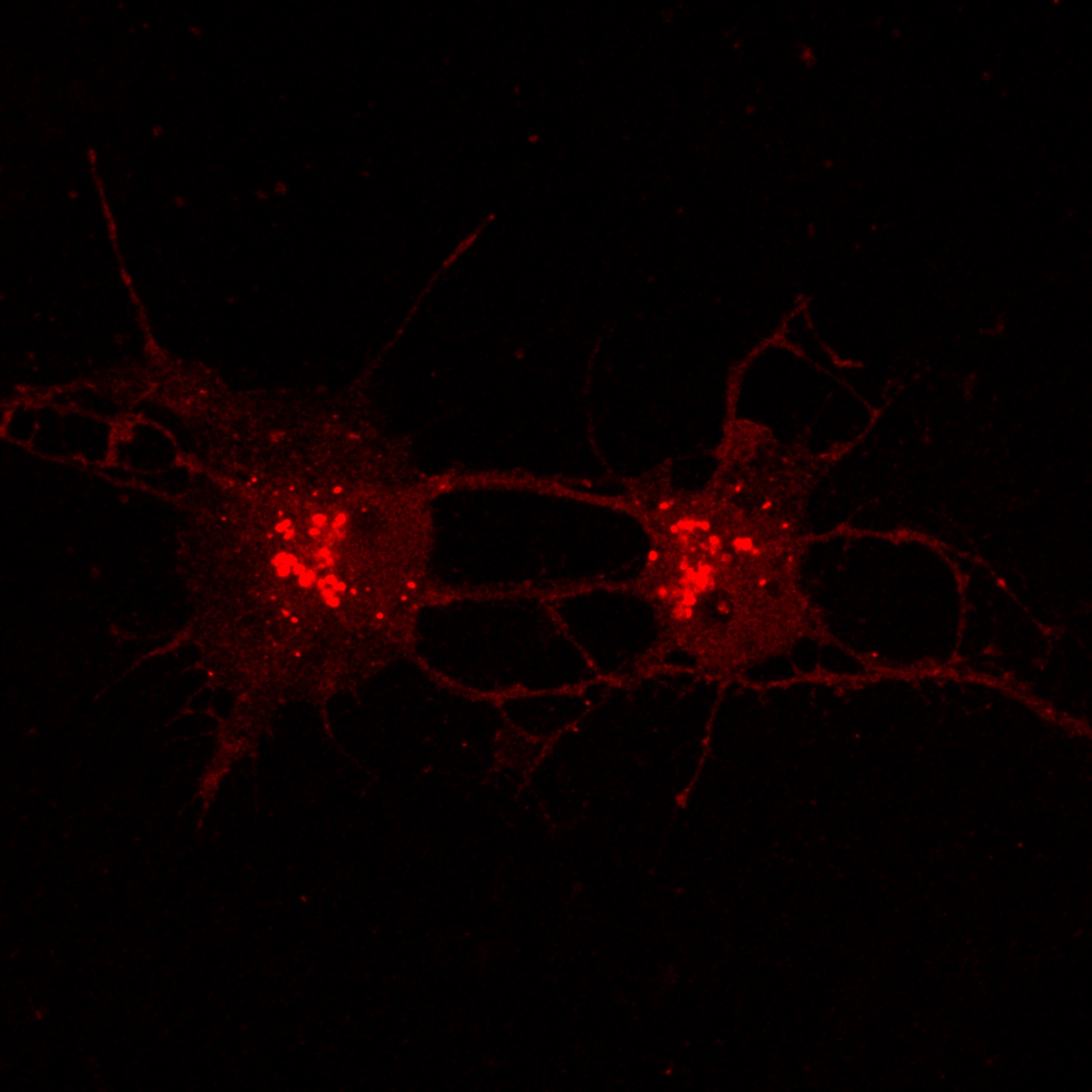
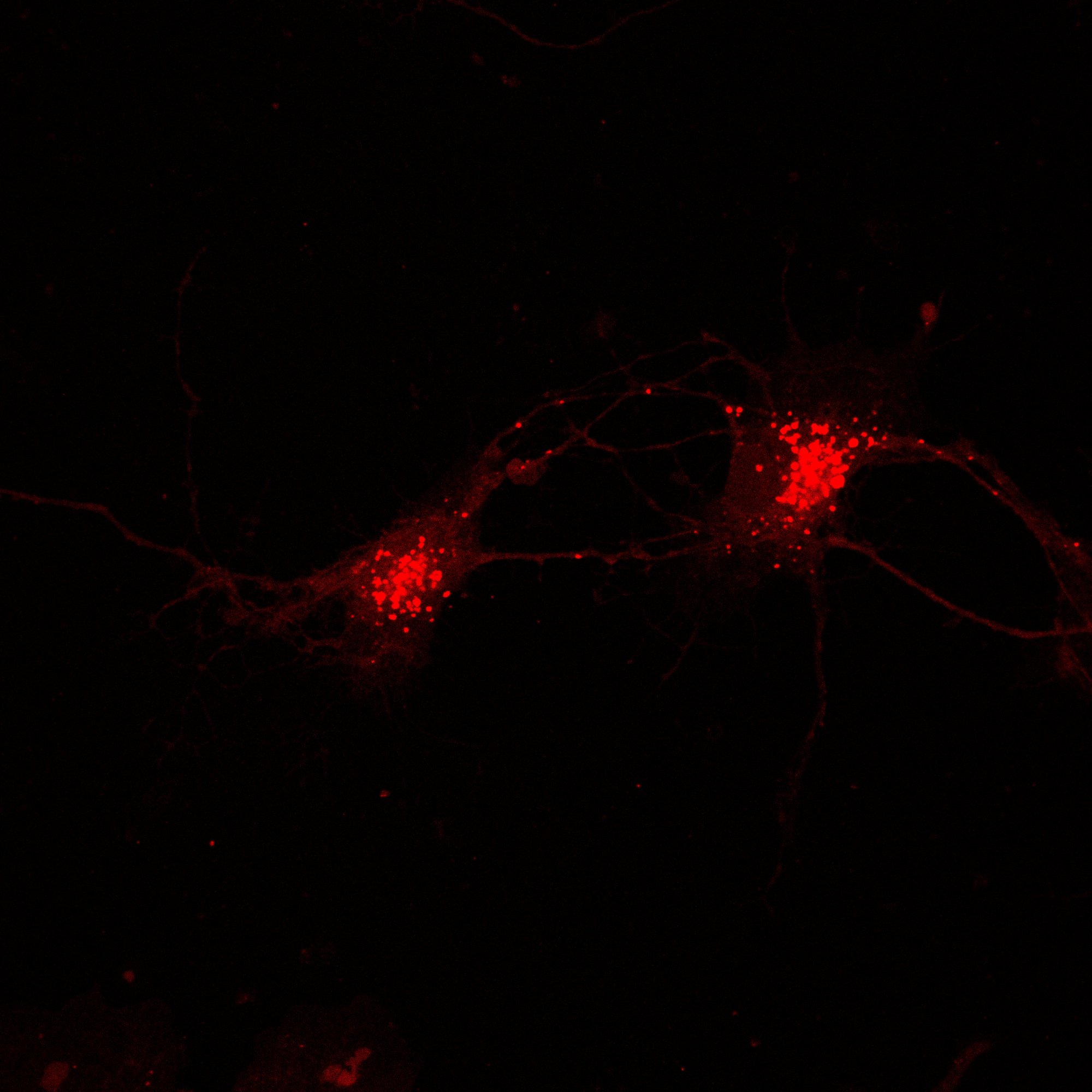
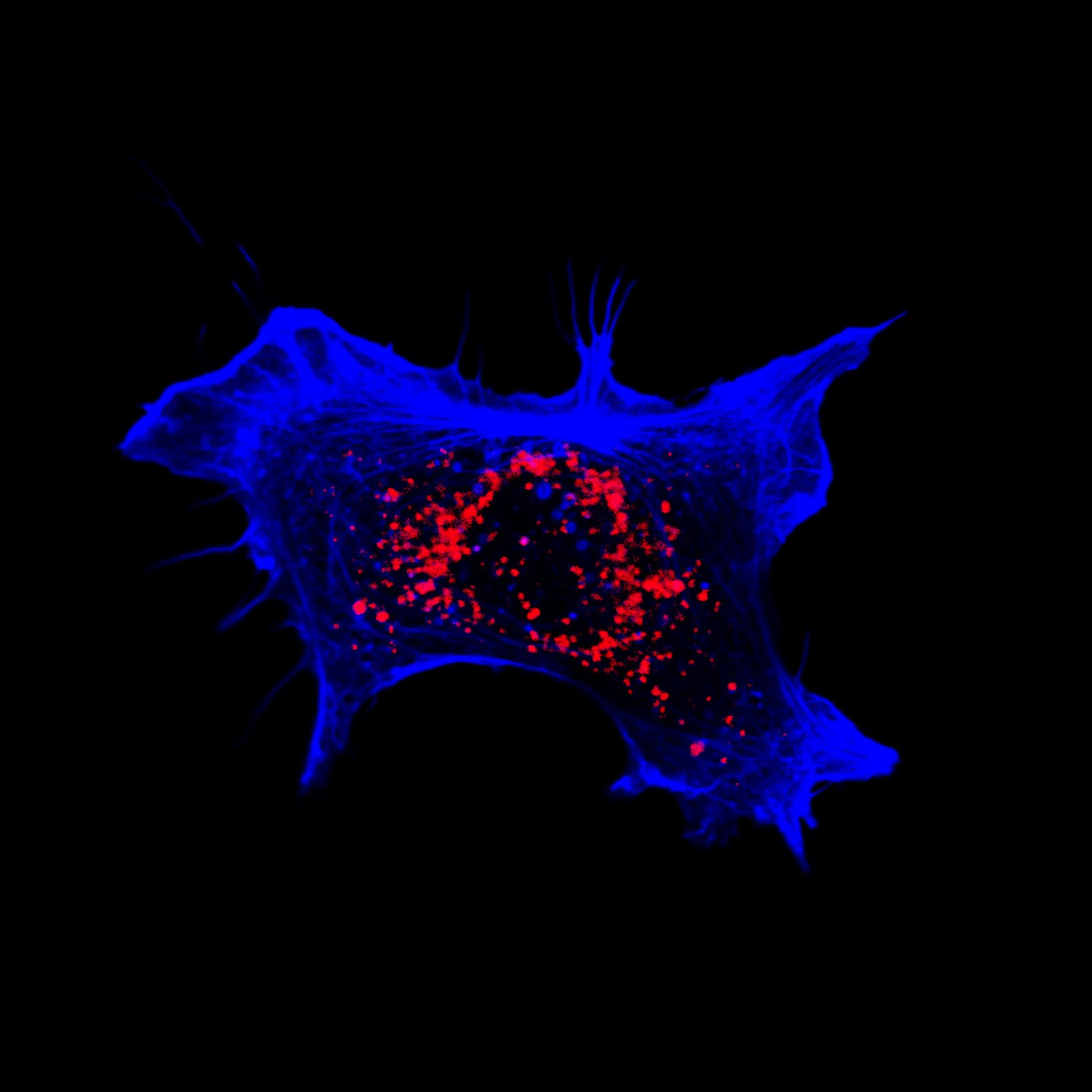
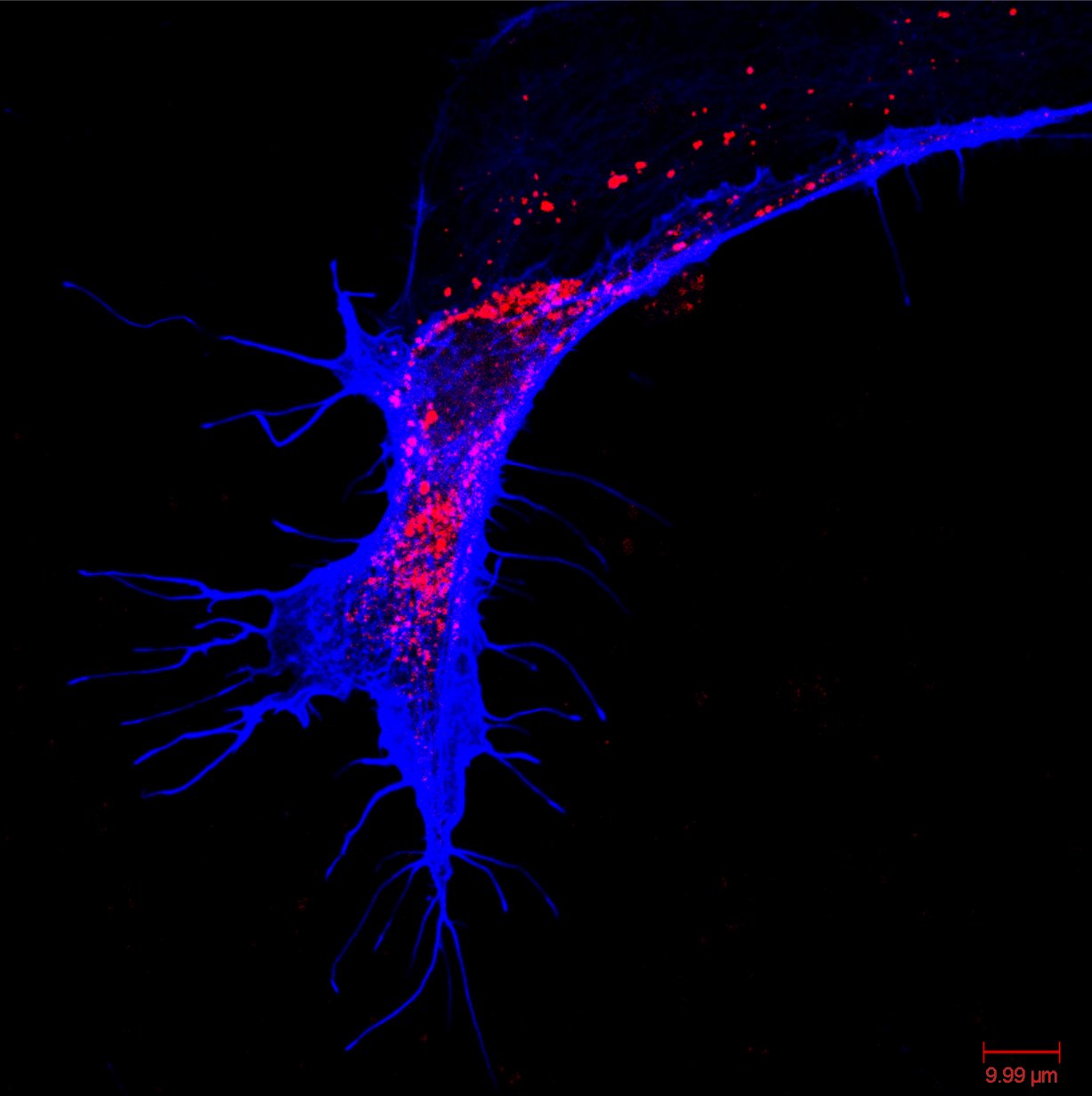
Hippocampal Dissection, Dissociation & Plating
Primary hippocampal neurons were isolated from E18 Sprague-Dawley rat embryos using established enzymatic dissociation protocols. Timed-pregnant dams were euthanized, embryos harvested under sterile conditions, and hippocampi dissected in ice-cold HBSS. Tissue was enzymatically digested with 0.25% trypsin-EDTA (15 min, 37°C), followed by mechanical trituration with fire-polished glass pipettes to generate a single-cell suspension.
Culture Protocol
Culture Yield & Viability
| Parameter | Value | Method |
|---|---|---|
| Yield per hippocampus | 1.2 ± 0.2 × 106 cells | Hemocytometer |
| Post-plating viability (4 h) | 94.2 ± 1.8% | Trypan blue |
| DIV7 viability | 91.6 ± 2.4% | Calcein-AM / PI |
| DIV14 viability | 88.3 ± 3.1% | Calcein-AM / PI |
| DIV21 viability | 82.7 ± 4.2% | Calcein-AM / PI |
| Neuronal purity (DIV7) | 92.4 ± 2.1% | MAP2+ / DAPI |
| Astrocyte contamination | 6.8 ± 1.4% | GFAP+ / DAPI |
| AraC treatment (DIV3) | 5 μM, 24 h | Anti-mitotic |