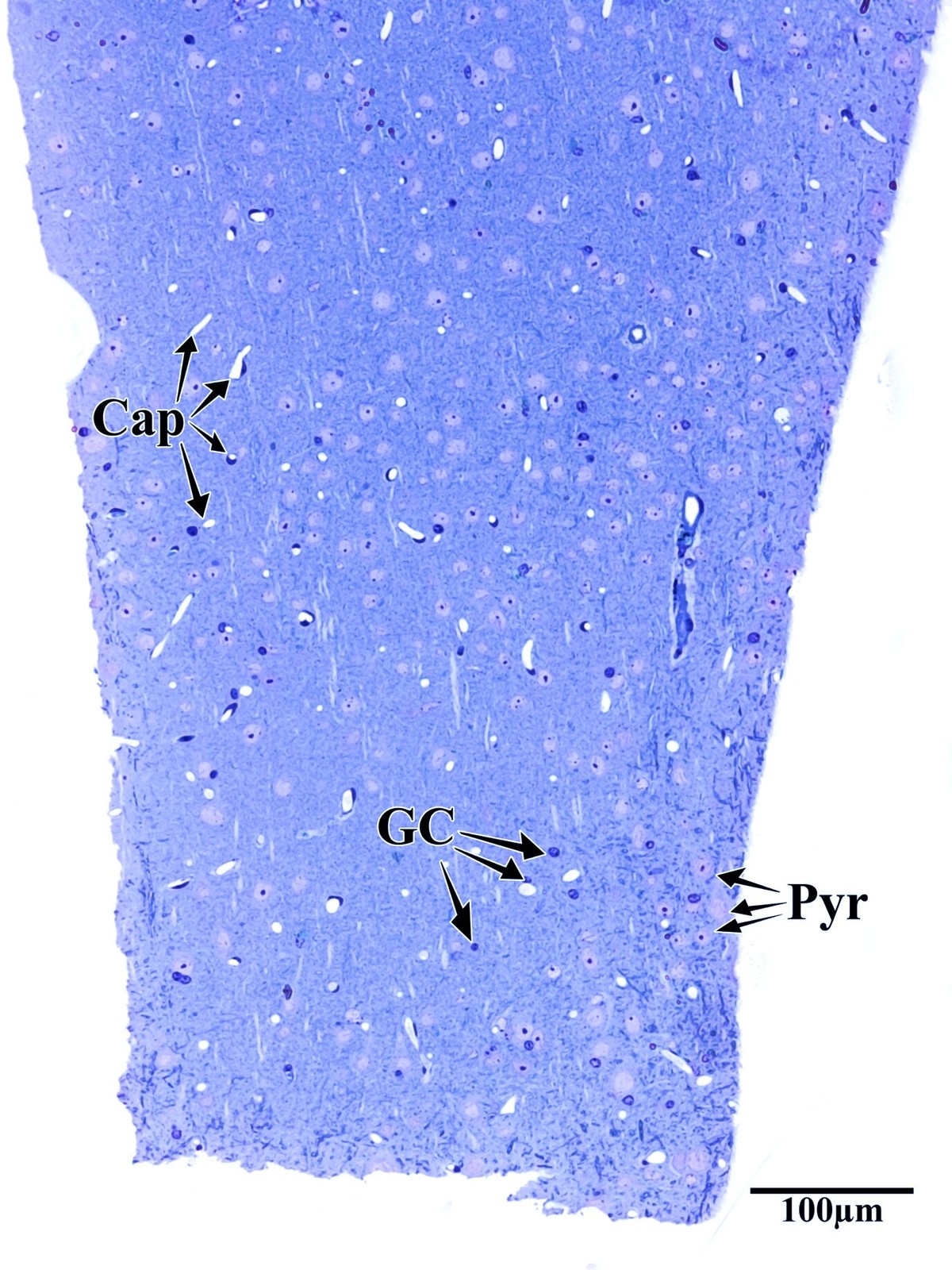
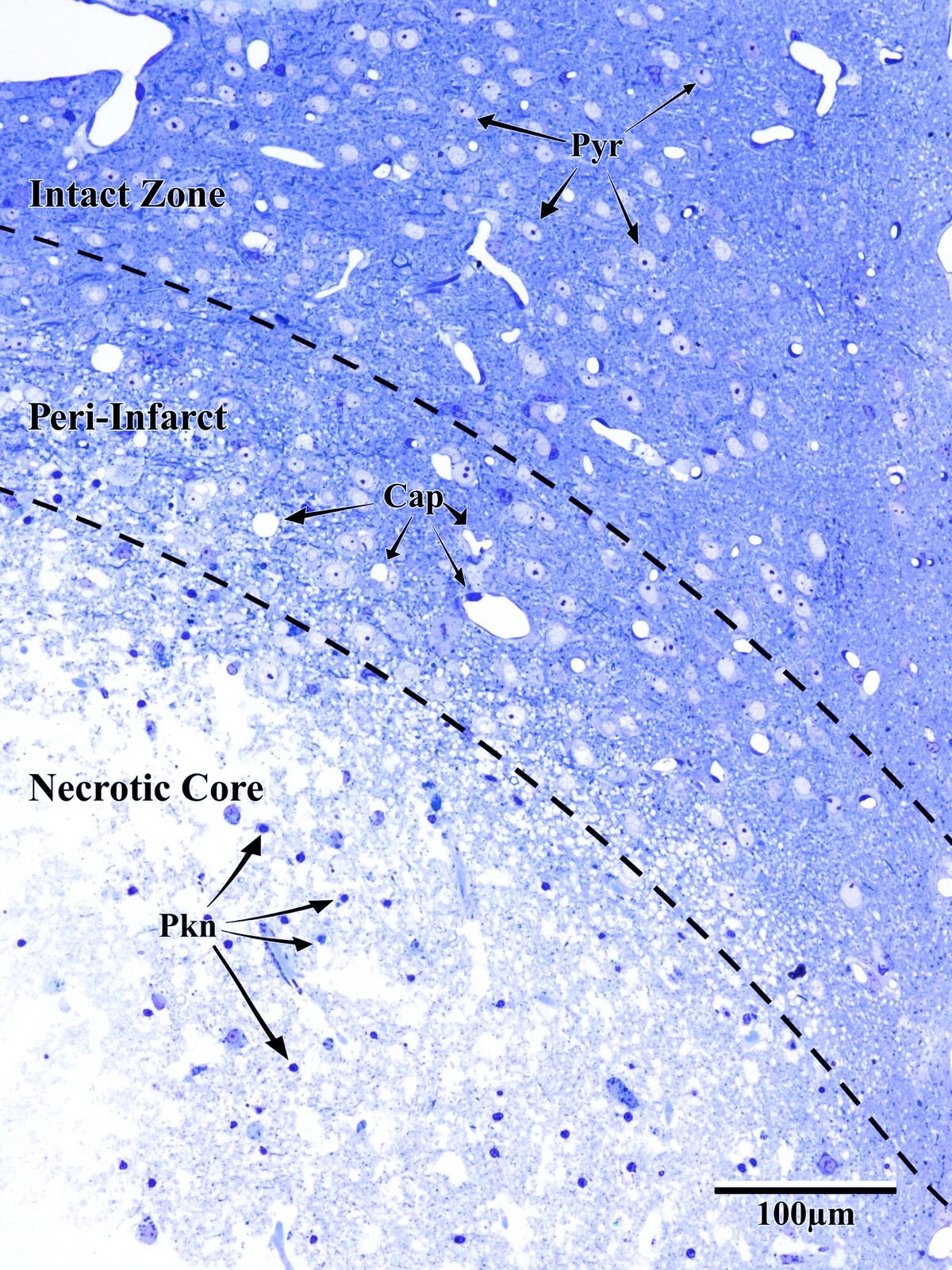
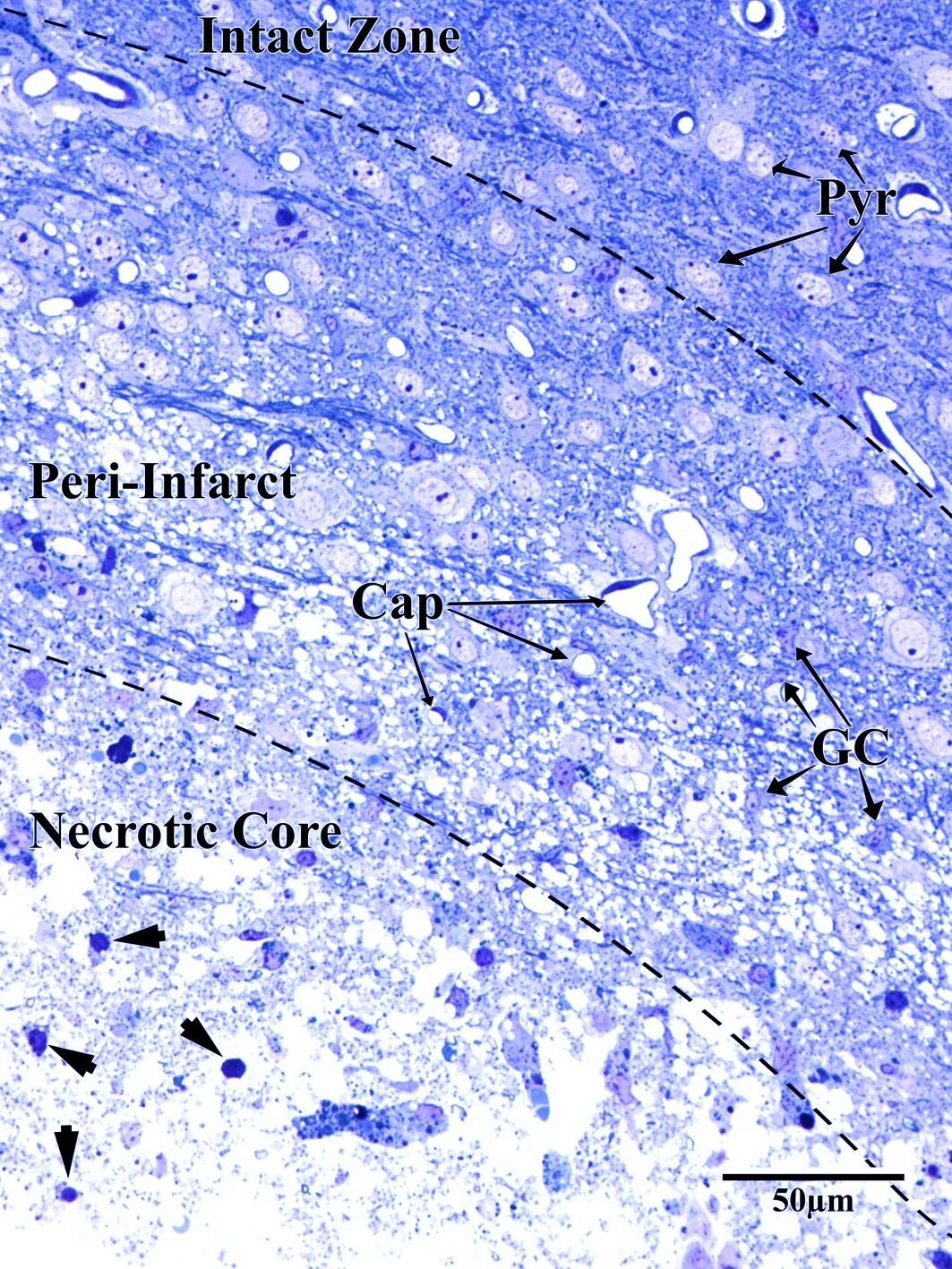
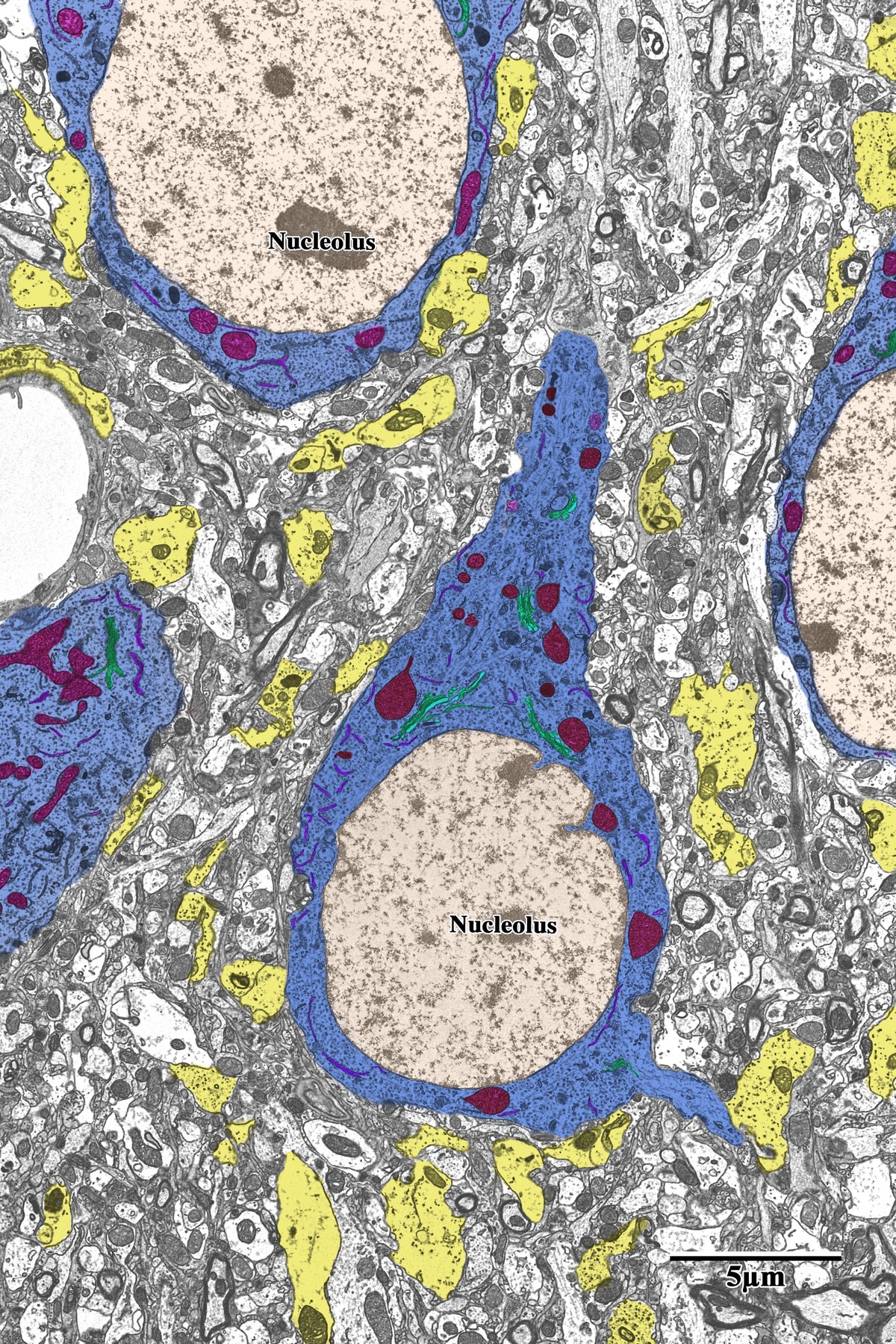
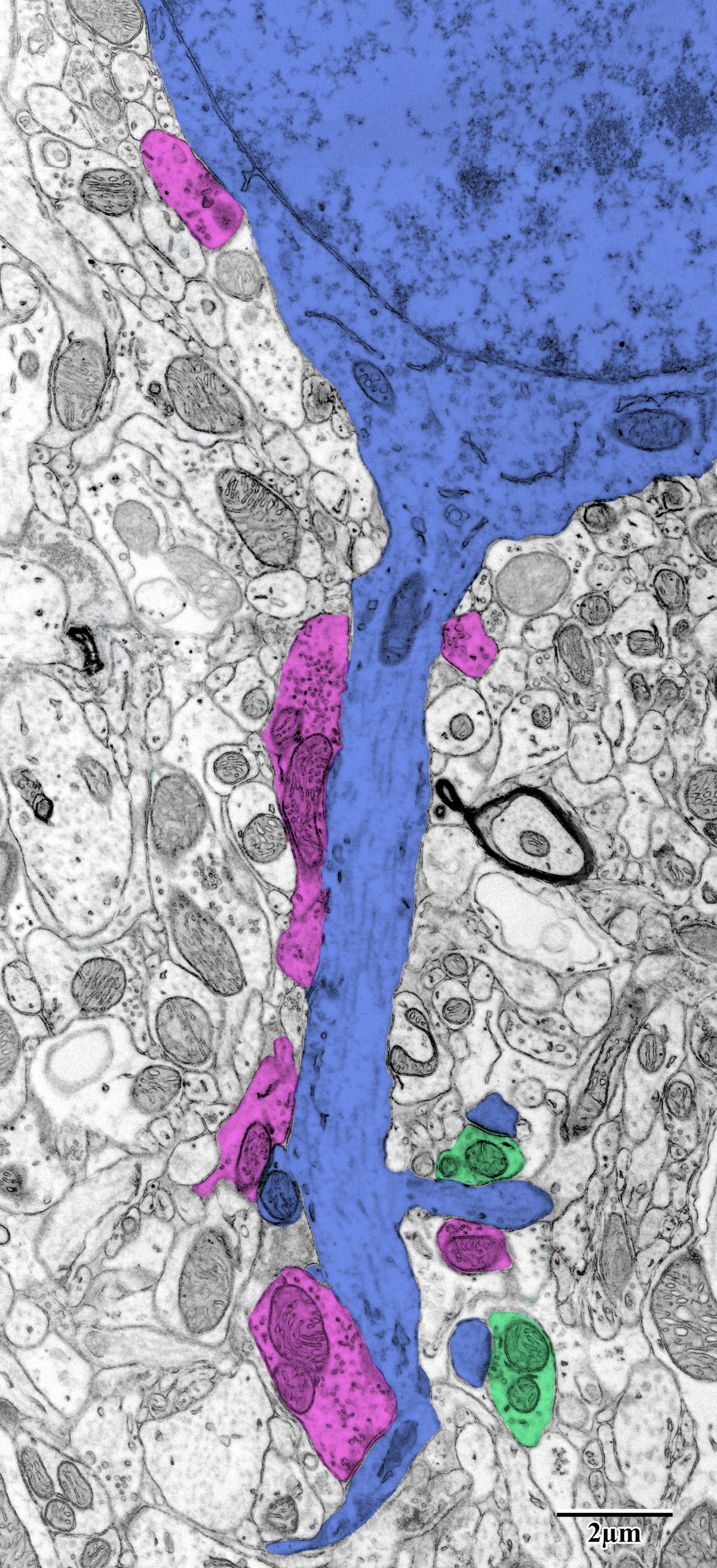
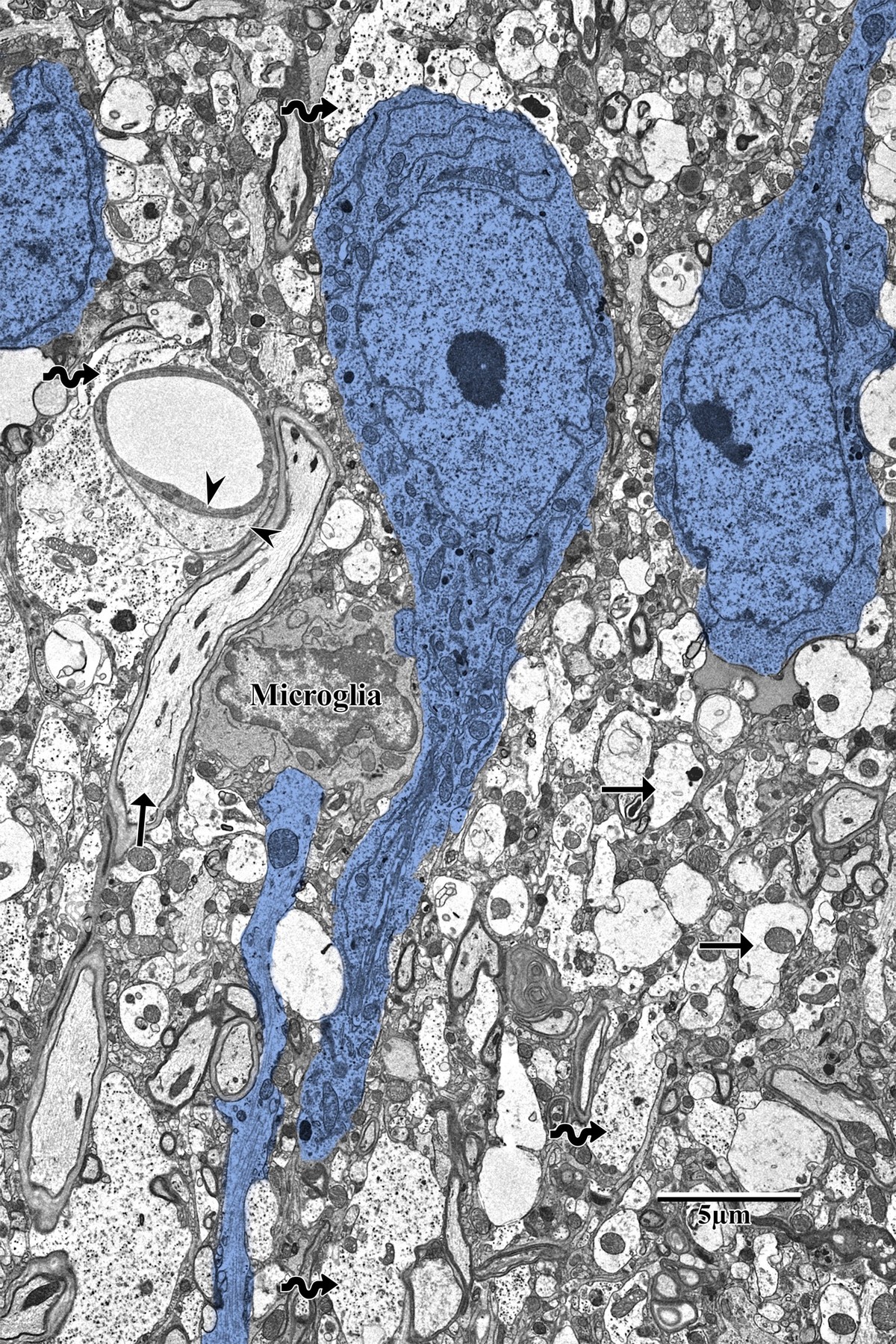
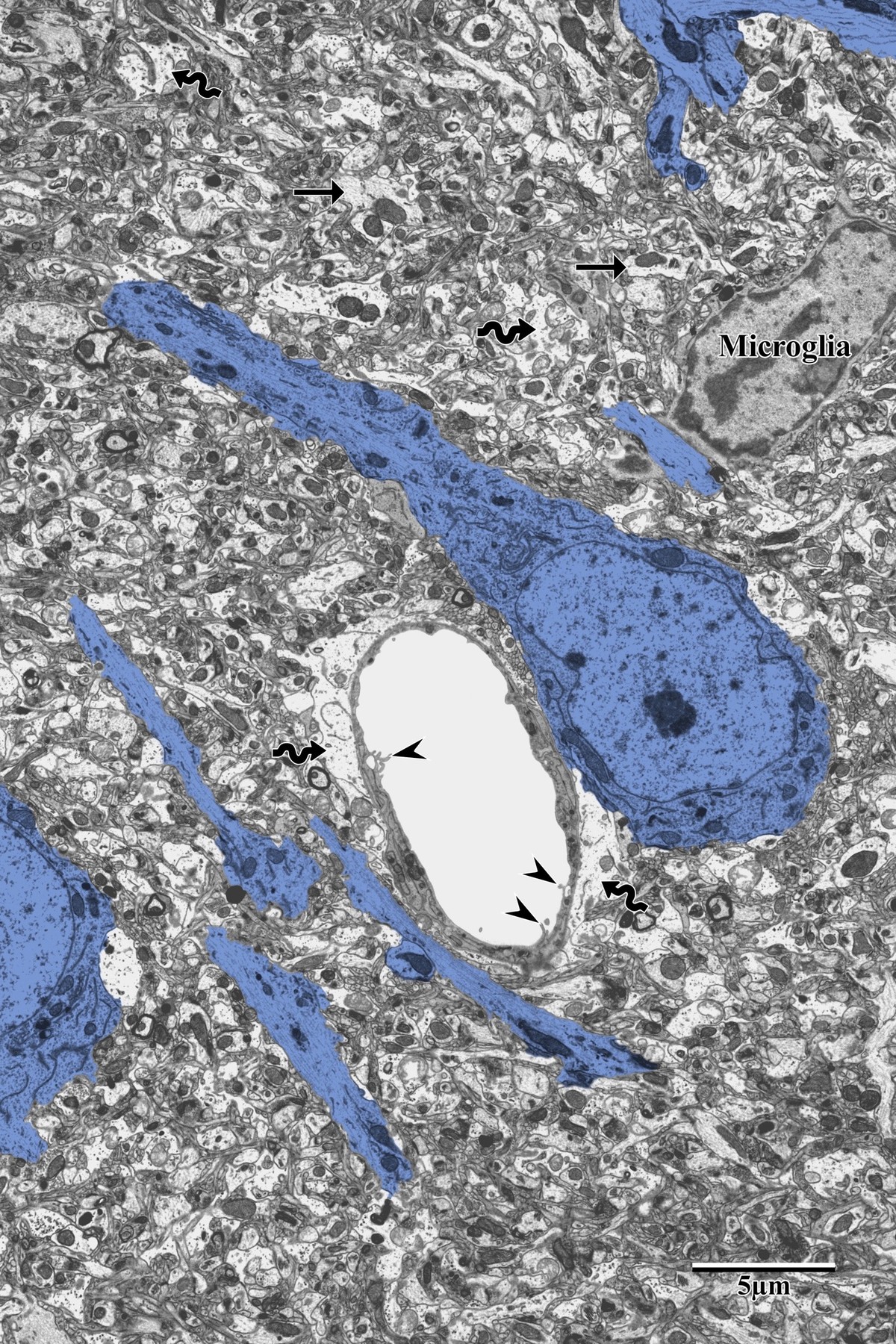
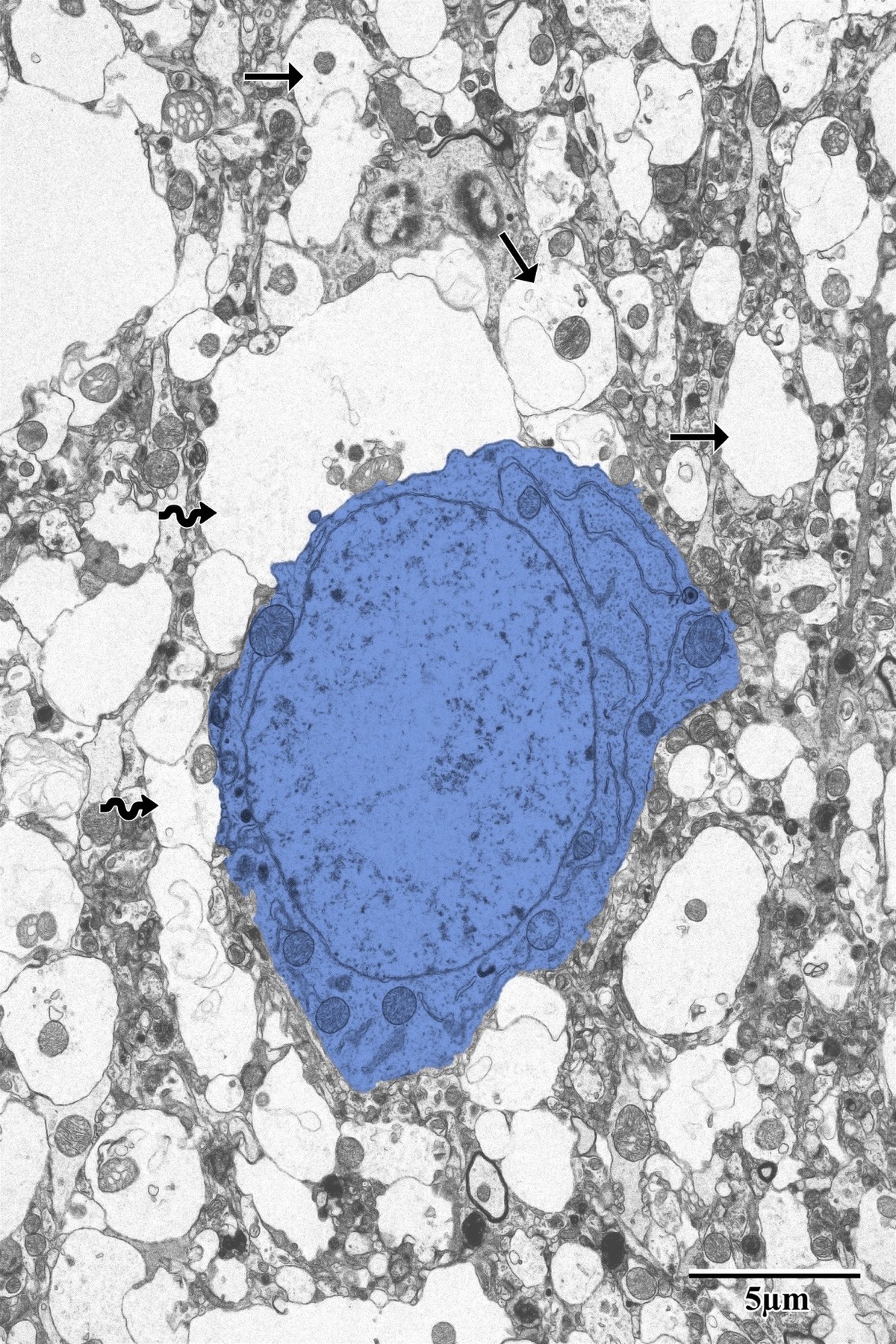
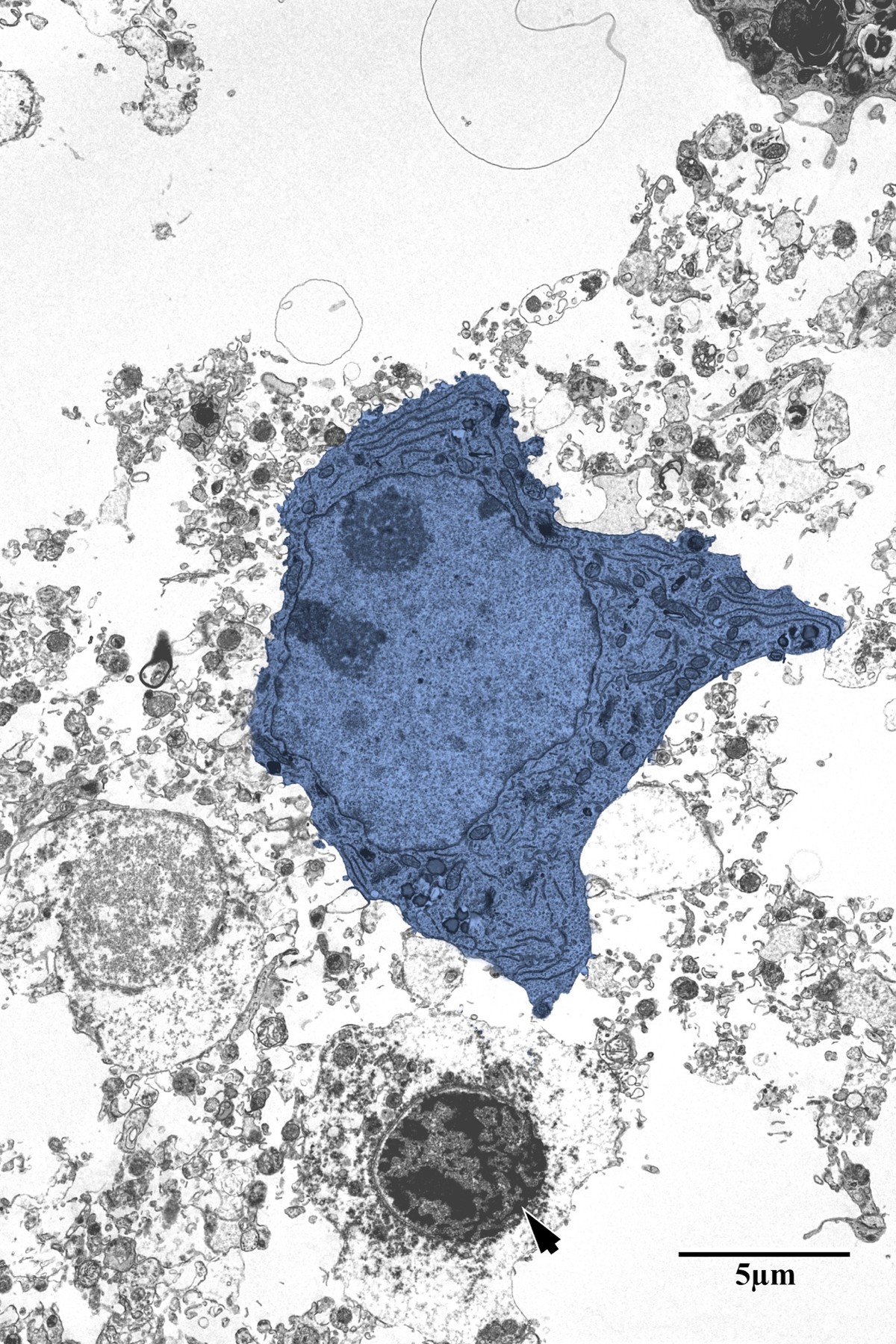
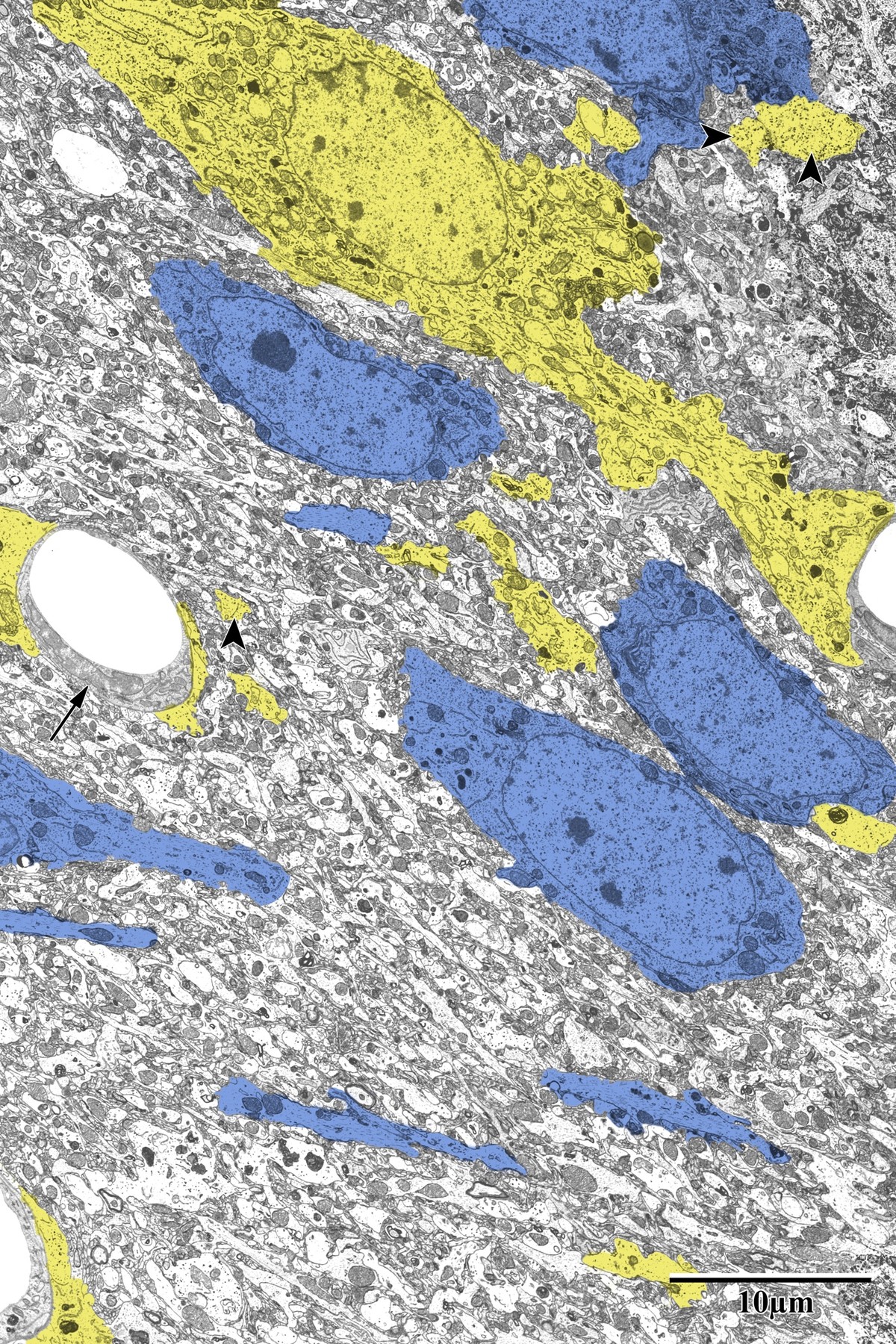
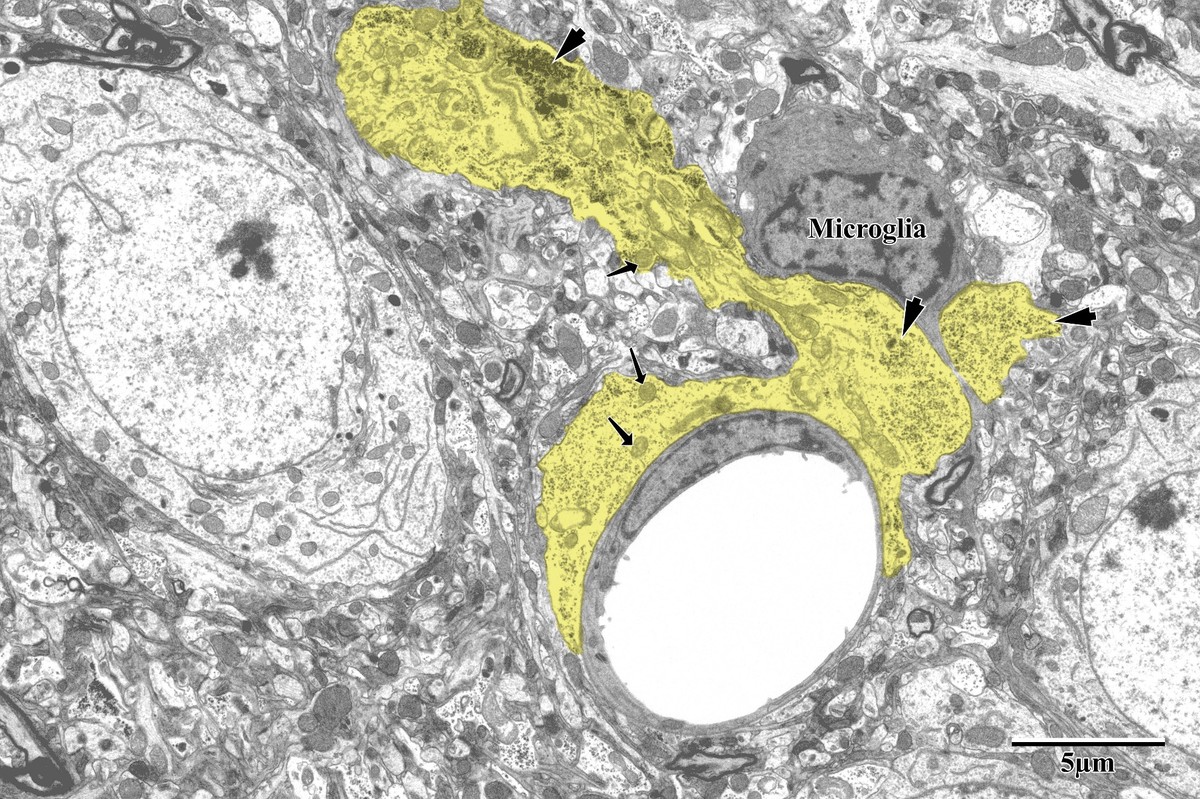
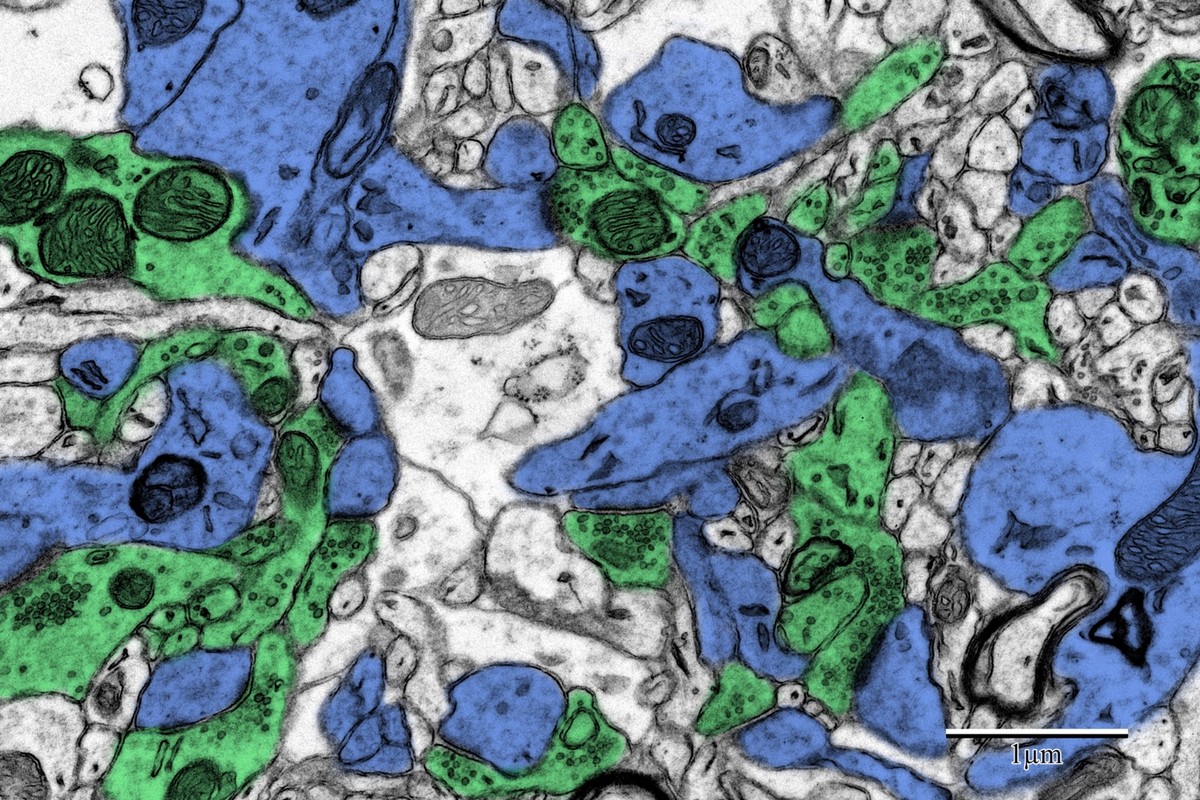
Photothrombotic Stroke Model
Focal unilateral stroke was induced in the somatosensory cortex (SSC) corresponding to the forelimb representation in adult mice (3 and 18 months, n = 5). Rose Bengal (110 mg/kg, IP) was administered as a photosensitizing agent, followed by targeted illumination with a green laser (532 nm, 17 mW, 15 min) through a thinned skull window. No craniotomy was needed — the skull was thinned to ~50% thickness with a dental drill under stereotaxic guidance.
Following 72 hours of recovery, mice were deeply anesthetized and prepared for transcardial perfusion fixation to preserve ultrastructure for both light and electron microscopy.